BIOtech Now
George Goodno

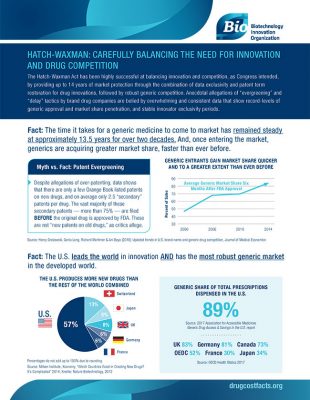
It is an underreported fact that the time it takes for a generic medicine to come to market has remained steady at approximately 13.5 years for over two decades. And, once entering the market, generics are acquiring greater market share, faster than ever before.
That’s why BIO is releasing its a new infographic, which highlights the highly successful effects of the Hatch-Waxman Act and the role it has played in balancing innovation and competition, as Congress intended.
BIO supports continued FDA initiatives to improve first-cycle generic approval rates, as this problem is the biggest barrier to more generic entry.
Powered by WPeMatico